Pixo Pods
Pixo Pods was recalled on 7 March 2025 under EU Safety Gate alert SR/00822/25. Chemical risk reported by Malta. The product is not equipped with a child-resistant fastening, which creates a health risk when in the hands of children.
| Alert Number | SR/00822/25 |
| Brand | Aspire |
| Category | Chemical products |
| Risk Type | Chemical |
| Notifying Country | Malta |
| Country of Origin | People's Republic of China |
| Published | 7 March 2025 |
Risk Description
The product is not equipped with a child-resistant fastening, which creates a health risk when in the hands of children. Moreover, the product has an excessive amount of nicotine-containing liquid (declared value: 3ml). This could lead to the accidental consumption of a high dose of nicotine. Nicotine is acutely toxic and can endanger the user’s safety, notably if the product comes into contact with the skin or is ingested. The product does not comply with the requirements of the Tobacco Products Directive.
Measures Taken
Type of economic operator to whom the measure(s) were ordered: OtherCategory of measure(s): Import rejected at borderDate of entry into force: 21/02/2025Type of economic operator taking notified measure(s): ManufacturerCategory of measure(s): Import rejected at borderDate of entry into force: 21/02/2025
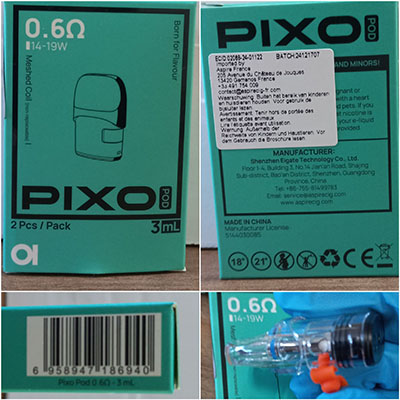
Product Description
Set consisting of two refillable cartridges, 3 mlResistance and power: 0.4 Ω: 23-30 W / 0.6 Ω: 14-19 W / 1.0 Ω: 10-15 W. Product sold online, in particular via gfc-provap.com.
🧪 What Should You Do?
This recall involves a chemical hazard related to Pixo Pods.
About this risk
This product contains prohibited or hazardous chemical substances that may cause skin irritation, allergic reactions, or long-term health effects.
Recommended action
Stop using the product immediately. Wash any skin that has come into contact with it. Keep it away from children. Return it to the retailer for a refund if possible.
Who is at risk?
Anyone who has used this product, especially children, pregnant women, and people with sensitive skin.