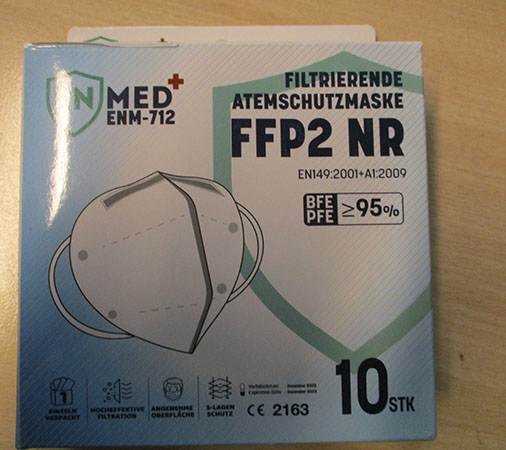
Filtrierende atemschutzmaske FFP2 NR
Filtrierende atemschutzmaske FFP2 NR was recalled on 24 December 2021 under EU Safety Gate alert A12/01835/21. Health risk / other risk reported by Belgium. The particle/filter retention of the material is insufficient (measured values: as low as 45%).
| Alert Number | A12/01835/21 |
| Brand | EN MeD+ |
| Category | Protective equipment |
| Risk Type | Health risk / other |
| Notifying Country | Belgium |
| Country of Origin | Türkiye |
| Published | 24 December 2021 |
Risk Description
The particle/filter retention of the material is insufficient (measured values: as low as 45%). Furthermore, weld points may tear apart generating holes in the mask, and ear straps may break easily. Consequently, the product does not fulfil the health and safety requirements; thus, even if combined with other recommended measures, it may not properly protect. The product does not comply with the Personal Protective Equipment (PPE) Regulation and with the European standard EN 149.
Measures Taken
Type of economic operator to whom the measure(s) were ordered: ImporterCategory of measure(s): Ban on the marketing of the product and any accompanying measuresDate of entry into force: Unknown
Product Description
White particle filter mask, marked with FFP2, and with the CE marking on the side.
🏥 What Should You Do?
This recall involves a health risk / other hazard related to Filtrierende atemschutzmaske FFP2 NR.
About this risk
This product poses a health risk that does not fall neatly into a single category. It may involve multiple hazards or an unusual safety concern identified by authorities.
Recommended action
Stop using the product and follow any specific instructions from the recall notice. Contact the manufacturer or your national consumer protection authority for more details.
Who is at risk?
All consumers who have purchased this product.