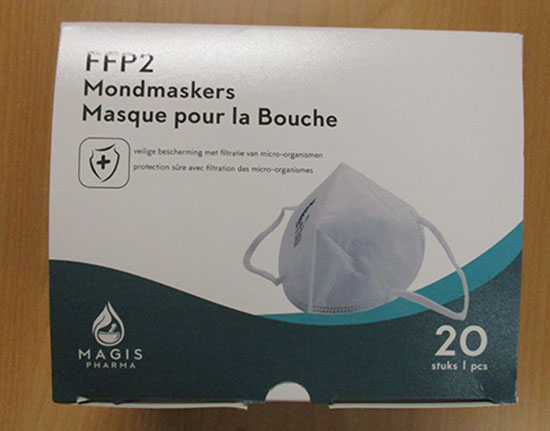
FFP2 Mondmaskers Masques pour la Bouche
Health risk / other Protective equipment Serious risk
| Alert Number | A12/01751/21 |
| Brand | Magis Pharma |
| Category | Protective equipment |
| Risk Type | Health risk / other |
| Notifying Country | Belgium |
| Country of Origin | People's Republic of China |
| Model Number | LK-008 |
| Published | 17 December 2021 |
Risk Description
The particle/filter retention of the material (measured values: as low as 92%) and the total filtration capacity of the mask (measured mean values: as low as 86%) are insufficient. Consequently, the product does not fulfil the health and safety requirements; thus, even if combined with other recommended measures, it may not properly protect. The product does not comply with the Personal Protective Equipment (PPE) Regulation and with the relevant European standard EN 149.
Measures Taken
Type of economic operator taking notified measure(s): ImporterCategory of measure(s): Recall of the product from end usersDate of entry into force: 20/11/2021
Product Description
Particle filter mask